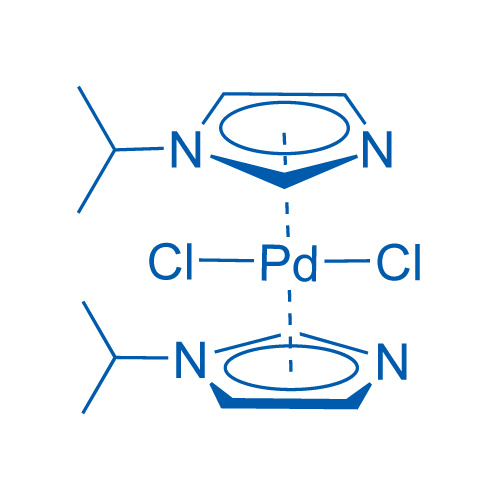
Structure, dynamics and catalytic activity of palladium(II) complexes with imidazole ligands - ScienceDirect

Polymer-supported palladium-imidazole complex catalyst for hydrogenation of substituted benzylideneanilines - ScienceDirect

Table 2 from SYNTHESIS AND STRUCTURE OF TWO PALLADIUM(II) COMPLEXES BEARING ACETONITRILE AND N-HETEROCYCLIC CARBENE DERIVED FROM IMIDAZOLE | Semantic Scholar
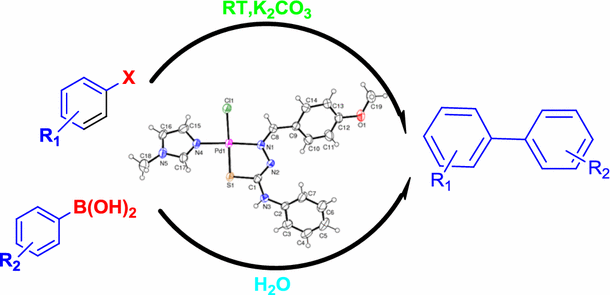
A thiosemicarbazone–palladium(II)–imidazole complex as an efficient pre-catalyst for Suzuki–Miyaura cross-coupling reactions at room temperature in aqueous media | SpringerLink
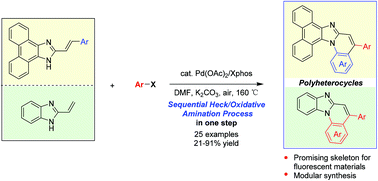
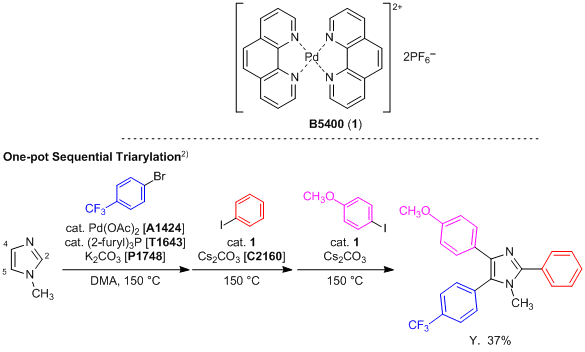
Palladium-catalyzed tandem one-pot synthesis of π-expanded imidazoles through a sequential Heck and oxidative amination reaction - Organic & Biomolecular Chemistry (RSC Publishing)

Structure, dynamics and catalytic activity of palladium(II) complexes with imidazole ligands - ScienceDirect
![NHC-palladium chloride-imidazole[NHC-Pd(II)-Im]complexes|1314876-23-7——Nanjing Finetech Chemical Co., Ltd. NHC-palladium chloride-imidazole[NHC-Pd(II)-Im]complexes|1314876-23-7——Nanjing Finetech Chemical Co., Ltd.](http://www.fine-chemtech.com/Uploads/5837a4c45b105.png)
NHC-palladium chloride-imidazole[NHC-Pd(II)-Im]complexes|1314876-23-7——Nanjing Finetech Chemical Co., Ltd.

Figure 2 from Self-assembled poly(imidazole-palladium): highly active, reusable catalyst at parts per million to parts per billion levels. | Semantic Scholar
![C(sp2)-H Functionalization of Imidazole at the C2- and C4-Position via Palladium-Catalyzed Isocyanide Insertion Leading to Indeno[1,2-d]imidazole and Imidazo[1,2-a]indole Derivatives.,The Journal of Organic Chemistry - X-MOL C(sp2)-H Functionalization of Imidazole at the C2- and C4-Position via Palladium-Catalyzed Isocyanide Insertion Leading to Indeno[1,2-d]imidazole and Imidazo[1,2-a]indole Derivatives.,The Journal of Organic Chemistry - X-MOL](https://xpic.x-mol.com/20200822%2F10.1021_acs.joc.0c01454.gif)
C(sp2)-H Functionalization of Imidazole at the C2- and C4-Position via Palladium-Catalyzed Isocyanide Insertion Leading to Indeno[1,2-d]imidazole and Imidazo[1,2-a]indole Derivatives.,The Journal of Organic Chemistry - X-MOL
![884879-23-6 | PHENYLALLYLCHLORO-[1,3-BIS(DIISOPROPYLPHENYL)IMIDAZOLE -2-YLIDENE]PALLADIUM(II) | Hangzhou Keying Chem Co., Ltd. 884879-23-6 | PHENYLALLYLCHLORO-[1,3-BIS(DIISOPROPYLPHENYL)IMIDAZOLE -2-YLIDENE]PALLADIUM(II) | Hangzhou Keying Chem Co., Ltd.](https://qncdn.chemcd.cn/keying/structure/KY155655.png)
884879-23-6 | PHENYLALLYLCHLORO-[1,3-BIS(DIISOPROPYLPHENYL)IMIDAZOLE -2-YLIDENE]PALLADIUM(II) | Hangzhou Keying Chem Co., Ltd.

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
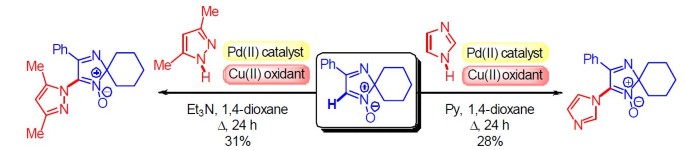
The First Example of Palladium(II)-Catalyzed Oxidative C–N Cross Coupling of 2 H -Imidazole 1-Oxide with Azoles | SpringerLink
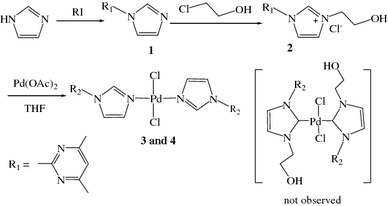
Synthesis, crystal structures, and catalytic activities of palladium imidazole complexes formed by 2-hydroxyethyl group cleavage | SpringerLink
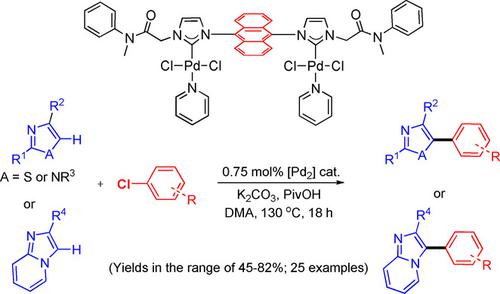
Dimetallic Palladium‐NHC Complexes: Synthesis, Characterization, and Catalytic Application for Direct C−H Arylation Reaction of Heteroaromatics with Aryl Chlorides - Adv. Synth. Catal. - X-MOL

Completely N1-selective palladium-catalyzed arylation of unsymmetric imidazoles: application to the synthesis of nilotinib. - Abstract - Europe PMC