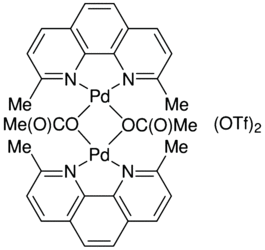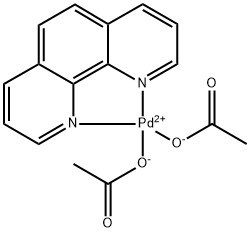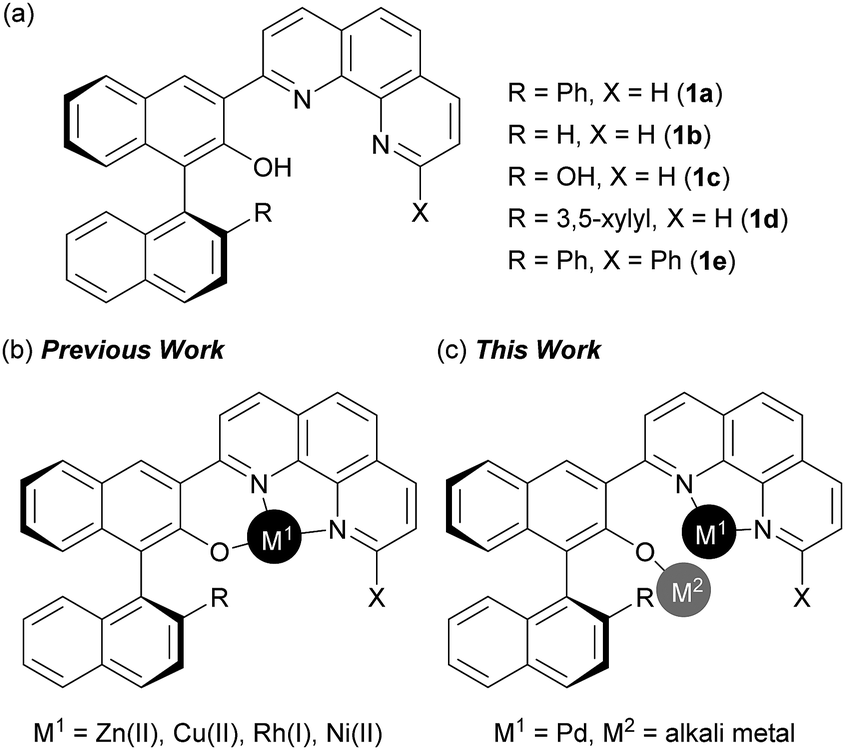
Design of bifunctional chiral phenanthroline ligand with Lewis basic site for palladium-catalyzed asymmetric allylic substitution - Chemical Communications (RSC Publishing) DOI:10.1039/C8CC00754C

Palladium‐Catalyzed Intramolecular Cyclization of Nitroalkenes: Synthesis of Thienopyrroles - El‐Atawy - 2017 - European Journal of Organic Chemistry - Wiley Online Library

Palladium-1,10-phenanthroline complex encaged in Y zeolite: An efficient and highly recyclable heterogeneous catalyst for aminocarbonylation - ScienceDirect
ChemIDplus - 132958-37-3 - QSAYSJLDPMTKTP-UHFFFAOYSA-L - 1,10-Phenanthroline-glycine palladium(II) - Similar structures search, synonyms, formulas, resource links, and other chemical information.
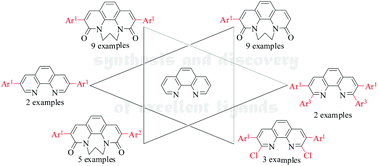
Direct arylation of phenanthroline derivatives via oxidative C–H/C–H cross-coupling: synthesis and discovery of excellent ligands - Organic & Biomolecular Chemistry (RSC Publishing)

The palladium–phenanthroline catalyzed carbonylation of nitroarenes to diarylureas: Effect of chloride and diphenylphosphinic acid - ScienceDirect

Scheme 1 Synthesis of phenanthroline-and Pd(II)-phenanthroline-based... | Download Scientific Diagram

A new method for the reaction of cross-coupling: preparation of 5,5′-bi(1,10-phenanthroline) in: Heterocyclic Communications Volume 23 Issue 1 (2017)

Design and synthesis of chiral 1,10-phenanthroline ligand, and application in palladium catalyzed asymmetric 1,4-addition reactions - ScienceDirect

Dichloro (1,10-phenanthroline-5,6-dione) palladium (II) complex supported by mesoporous silica SBA-15 as a photocatalyst for degradation of 2,4-dichlorophenol - ScienceDirect

Design and synthesis of chiral 1,10-phenanthroline ligand, and application in palladium catalyzed asymmetric 1,4-addition reactions - ScienceDirect

PDF) Friedel–Crafts-Type Alkylation of Indoles in Water Using Amphiphilic Resin-Supported 1,10-Phenanthroline–Palladium Complex under Aerobic Conditions