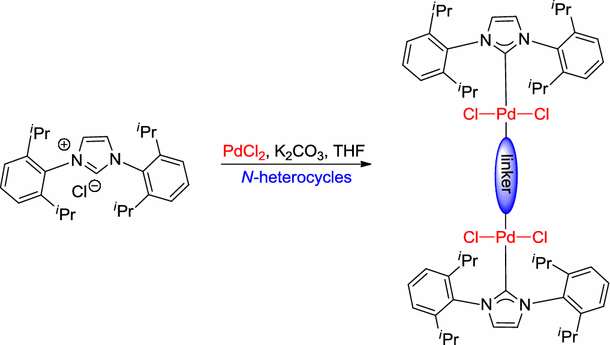
Scheme 1 | Dinuclear NHC–palladium(II) complexes: synthesis,characterization and application to Suzuki–Miyaura cross-coupling reactions | SpringerLink
![EP2800746B1 - Process for the manufacture of 1-[2-(2,4-dimethyl-phenylsulfanyl)-phenyl]-piperazine - Google Patents EP2800746B1 - Process for the manufacture of 1-[2-(2,4-dimethyl-phenylsulfanyl)-phenyl]-piperazine - Google Patents](https://patentimages.storage.googleapis.com/0a/c0/98/435d1a40b7cf08/imgb0013.png)
EP2800746B1 - Process for the manufacture of 1-[2-(2,4-dimethyl-phenylsulfanyl)-phenyl]-piperazine - Google Patents

Palladium supported on modified magnetic nanoparticles: a phosphine‐free and heterogeneous catalyst for Suzuki and Stille reactions - Ghorbani‐Choghamarani - 2016 - Applied Organometallic Chemistry - Wiley Online Library
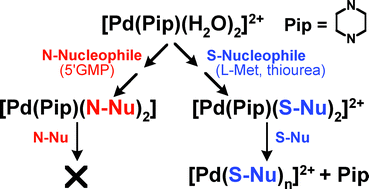
Kinetics and mechanism of the substitution behaviour of Pd(ii) piperazine complexes with different biologically relevant nucleophiles - Dalton Transactions (RSC Publishing)
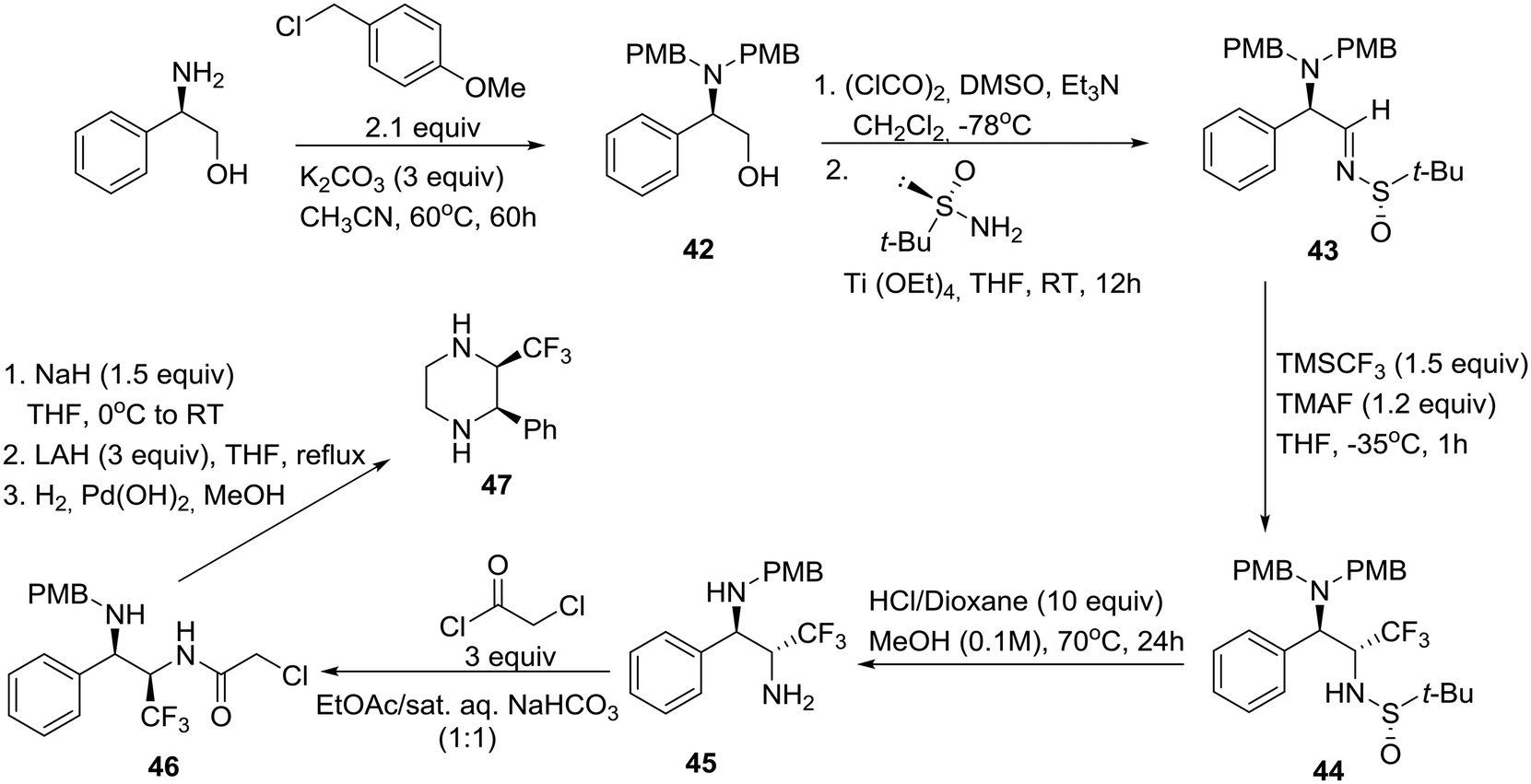
Recent progress toward the asymmetric synthesis of carbon-substituted piperazine pharmacophores and oxidative related heterocycles - RSC Medicinal Chemistry (RSC Publishing) DOI:10.1039/D0MD00053A

Modification of silica using piperazine for immobilization of palladium nanoparticles: a study of its catalytic activity as an efficient heterogeneous catalyst for Heck and Suzuki reactions | SpringerLink
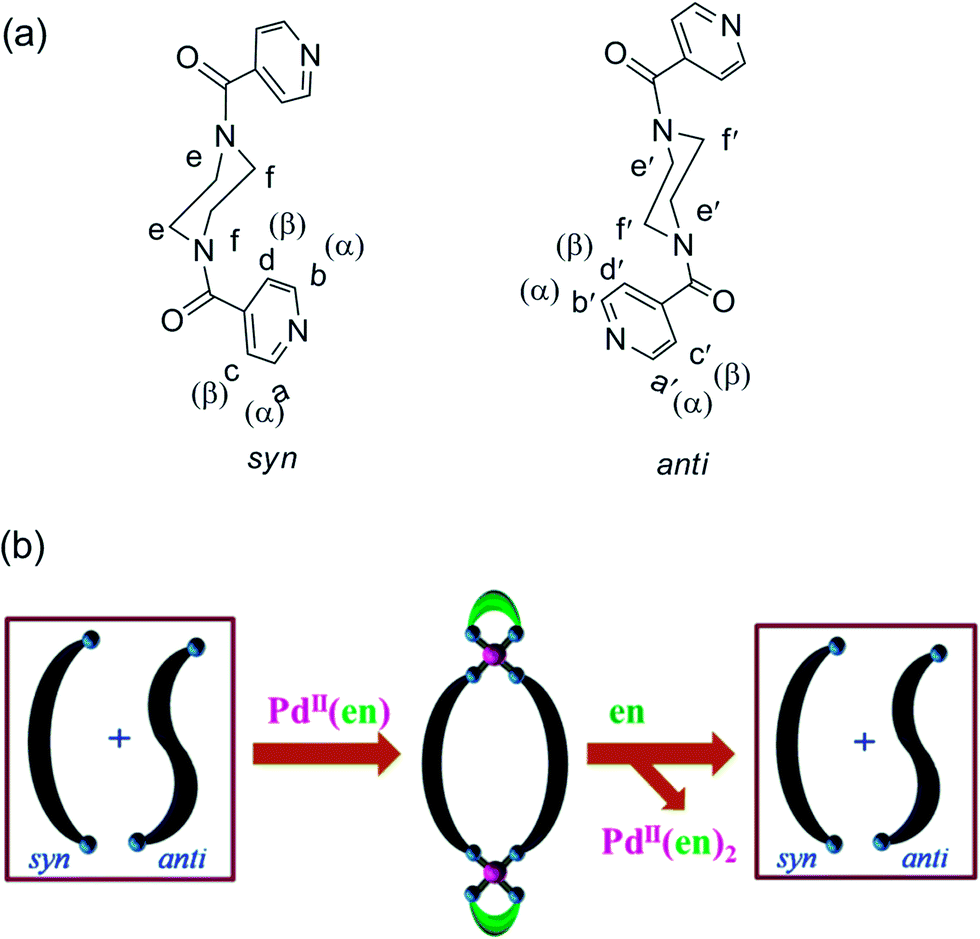
Palladium( ii ) induced complete conformational enrichment of the syn isomer of N , N ′-bis(4-pyridylformyl)piperazine - RSC Advances (RSC Publishing) DOI:10.1039/C3RA47904H
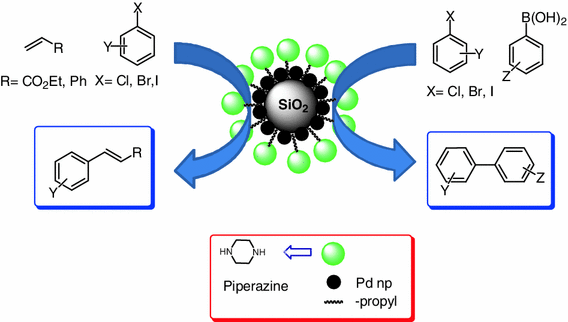
Modification of silica using piperazine for immobilization of palladium nanoparticles: a study of its catalytic activity as an efficient heterogeneous catalyst for Heck and Suzuki reactions | SpringerLink

Synthesis of N-arylpiperazines from aryl halides and piperazine under a palladium tri-tert-butylphosphine catalyst - ScienceDirect
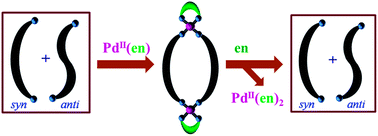
Palladium(ii) induced complete conformational enrichment of the syn isomer of N,N′-bis(4-pyridylformyl)piperazine - RSC Advances (RSC Publishing)
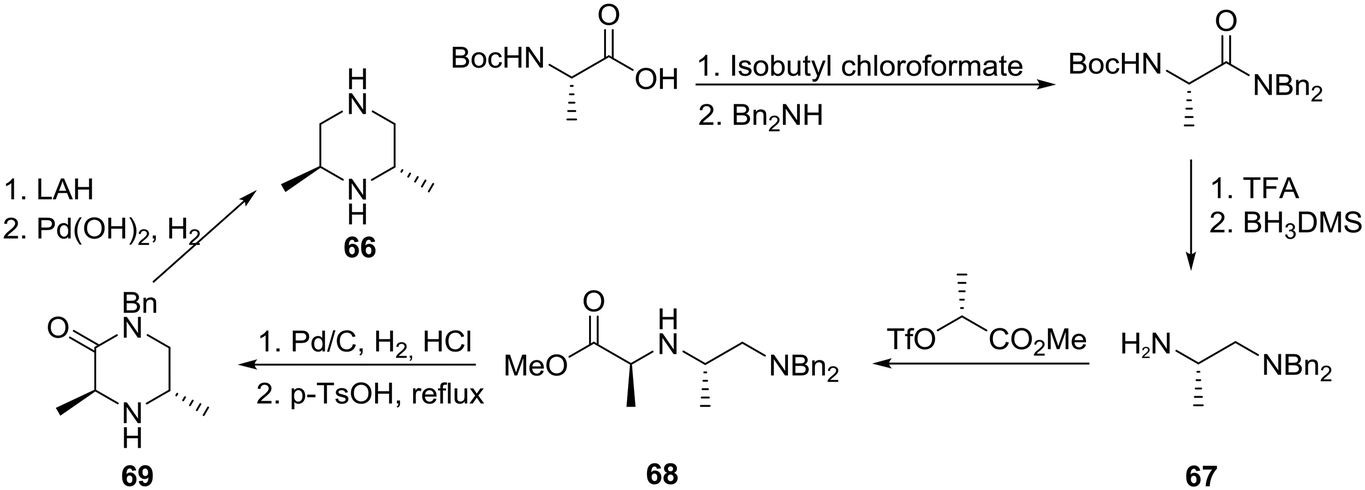
Recent progress toward the asymmetric synthesis of carbon-substituted piperazine pharmacophores and oxidative related heterocycles - RSC Medicinal Chemistry (RSC Publishing) DOI:10.1039/D0MD00053A
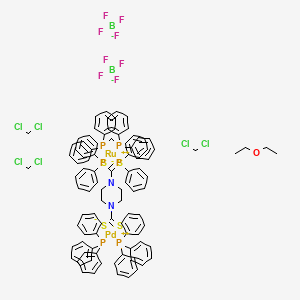
Dichloromethane;diphenylphosphanylmethyl(diphenyl)phosphane;ethoxyethane; palladium(2+);piperazine-1,4-dicarbodithioate;ruthenium(2+);triphenylphosphane;ditetrafluoroborate | C99H98B2Cl6F8N2OP6PdRuS4 - PubChem

Piperazine as an Inexpensive and Efficient Ligand for Pd-Catalyzed Homocoupling Reactions to Synthesize Bipyridines and Their Analogues | Bentham Science
![A practical chromatography-free synthesis of a 5,6-dihydroimidazolo[1,5-f]pteridine derivative as a polo-like kinase-1 inhibitor - Tetrahedron - X-MOL A practical chromatography-free synthesis of a 5,6-dihydroimidazolo[1,5-f]pteridine derivative as a polo-like kinase-1 inhibitor - Tetrahedron - X-MOL](https://xpic.x-mol.com/20180817%2F10.1016_j.tet.2018.08.020.jpg)
A practical chromatography-free synthesis of a 5,6-dihydroimidazolo[1,5-f]pteridine derivative as a polo-like kinase-1 inhibitor - Tetrahedron - X-MOL

Synthesis of 2,6-Disubstituted Piperazines by a Diastereoselective Palladium-Catalyzed Hydroamination Reaction

Table 2 from Kinetics and mechanism for the substitution reactions of monoaquamonochloro-(piperazine) palladium (II) complex with L-methionine and thiourea in aqueous solution | Semantic Scholar
![EP2800746B1 - Process for the manufacture of 1-[2-(2,4-dimethyl-phenylsulfanyl)-phenyl]-piperazine - Google Patents EP2800746B1 - Process for the manufacture of 1-[2-(2,4-dimethyl-phenylsulfanyl)-phenyl]-piperazine - Google Patents](https://patentimages.storage.googleapis.com/53/5f/9b/46ea6342cf2651/imgb0001.png)
EP2800746B1 - Process for the manufacture of 1-[2-(2,4-dimethyl-phenylsulfanyl)-phenyl]-piperazine - Google Patents

Synthesis of arylpiperazines via palladium-catalyzed aromatic amination reaction with unprotected piperazines - ScienceDirect

Pd-Catalyzed Synthesis of Piperazine Scaffolds Under Aerobic and Solvent-Free Conditions. - Abstract - Europe PMC
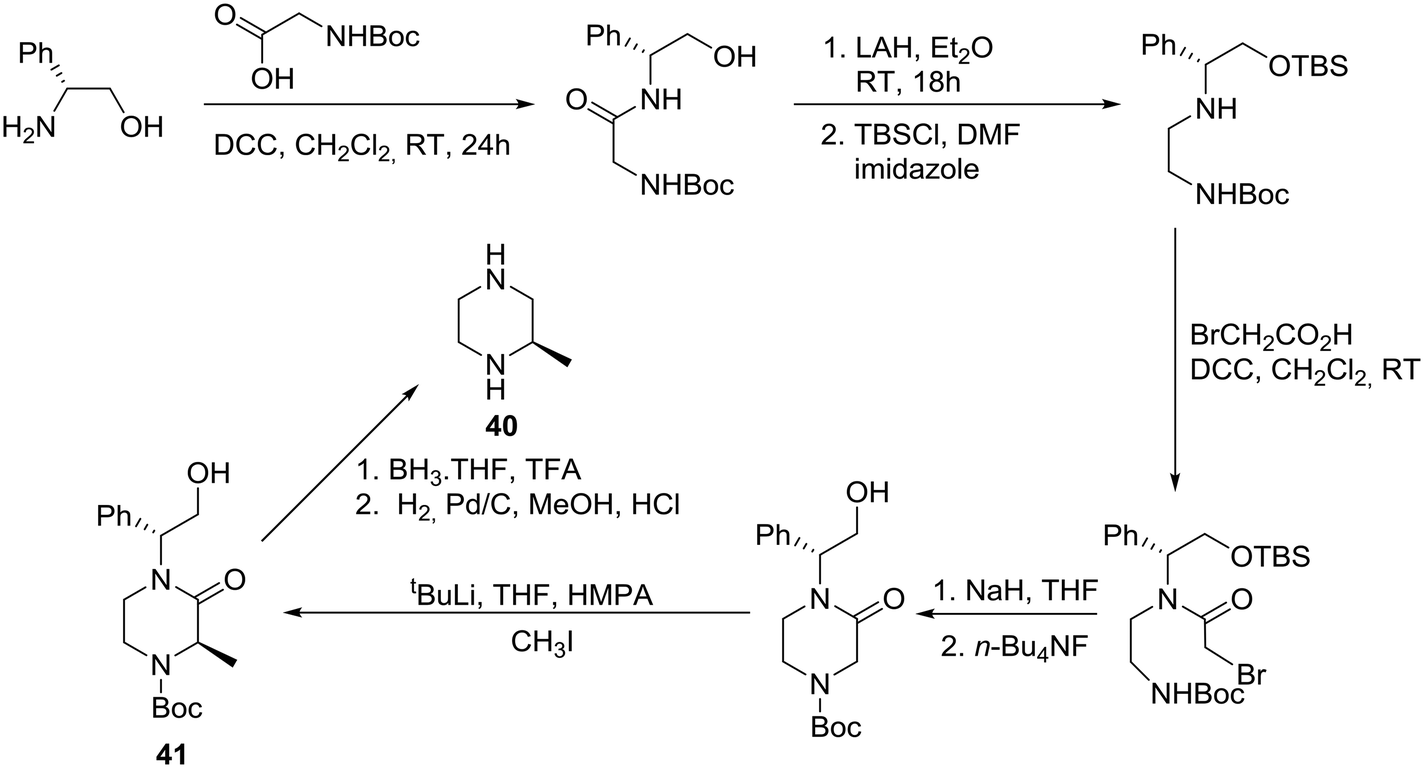
Recent progress toward the asymmetric synthesis of carbon-substituted piperazine pharmacophores and oxidative related heterocycles - RSC Medicinal Chemistry (RSC Publishing) DOI:10.1039/D0MD00053A


